|
12/8/2023 0 Comments Chromium 58 proton number 
SIDE NOTE Actually, chromium-51 decays via electron capture to vanadium-51, so I assume that this was just a practice problem for understanding how to balance nuclear equations and how various types of radioactive decay work. Science Chemistry Isotope How many protons electrons and neutrons does chromium-58 have Question: How many protons electrons and neutrons does chromium-58 have Chromium: Chromium. The mass of the longest lived isotope is given for elements without a. The radioactive decay that in which an electron and an electron antineutrino are emitted from the nucleus of a radioactive isotope is called beta-minus decay, #beta^-# decay. This table lists the mass and percent natural abundance for the stable nuclides. So, the nuclear equation will look like this In addition to this, in order for the charge to be preserved as well, an electron antineutrino, #bar(nu)_e#. Well, if a neutron is converted into a proton, the mass number will remain unchanged, but the atomic number will increase by #1#. 
This means that a neutron from chromium-51's nucleus was converted into a proton and an electron. Notice that the product of the decay has one extra proton than chromium-51, but that their mass numbers are equal. 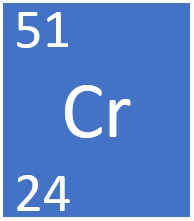
So, what must happen in order for this reaction to take place? Chromium-58 Chromium-63 of protons of neutrons Fluorine (F atomic number 9) has 7. For the following Chromium isotopes, write the number of protons and neutrons. The nuclear equation for this decay will look like this Chromium in general is symbolised as: 24Cr52 / Chromium-52 Chromium has an atomic number of 24 which is equal to the number of prot. However, since you're essentially dealing with the isotopes of the different elements, chromium and manganese, then you know for a fact that their atomic numbers will be different.Ī quick look in the periodic table will show you that chromium's atomic number is equal to #24# and manganese's atomic number is equal to #25#. In your case, you know that you're dealing with two isotopes that have the same mass number, #51#. So, you know that chromium-51 decays into manganese-51, but you don't know what type of radiation is emitted when this takes place.įor an isotope, the number that follows the element's name represents its mass number, which tells you how many protons and neutrons the isotope has in its nucleus. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
